
Biospace Lab provides innovative optical and autoradiography preclinical imaging systems to help worldwide researchers push the boundaries of their biological studies.
bioluminescence & fluorescence
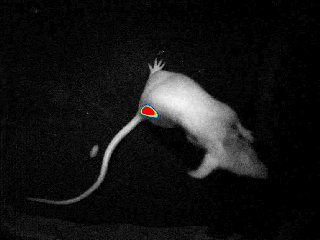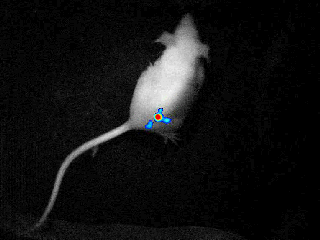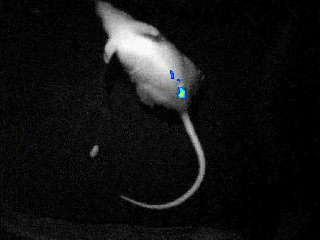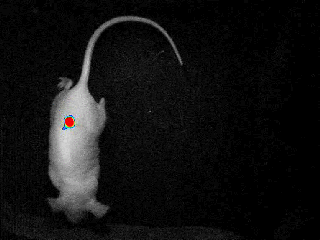
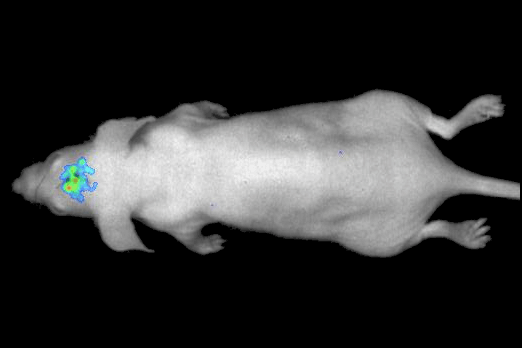
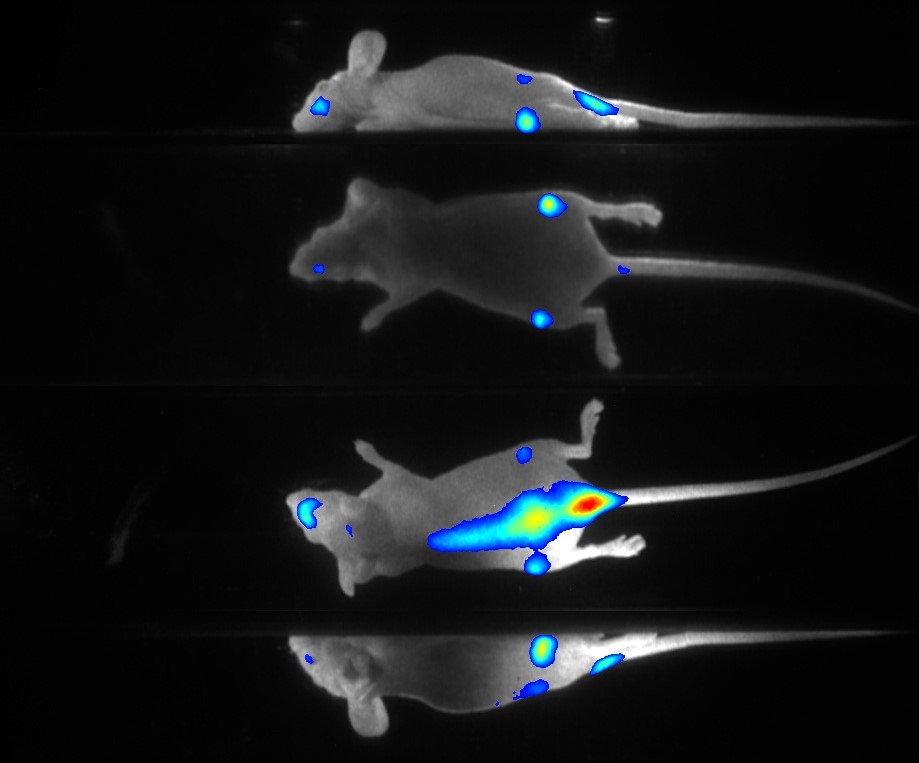
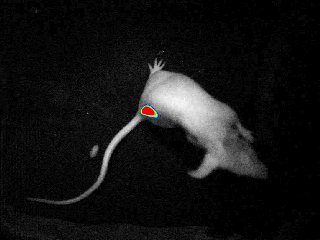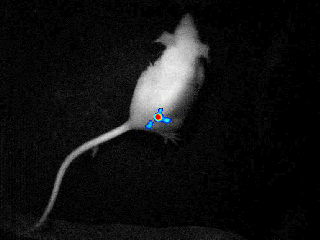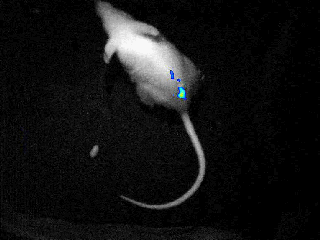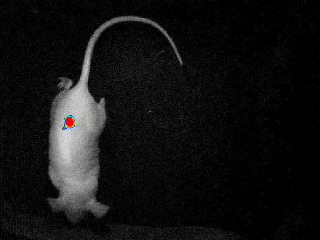
Our real-time optical imaging systems enable you to see different types of complex biological processes by combining bioluminescence, fluorescence, X-ray imaging technologies in a single instrument.
Thanks to our modular approach, your specific requirements for optimized localization, resolution and visualization can be easily accommodated by adding 3D, macrolens, in action, X-ray options.
As a pioneer in imaging of non-anesthetized animals, Biospace Lab provides a unique solution to perform your experimentations on free-moving animals in physiological conditions.
With our systems, you will be able to:
- Detect the tiniest signals from a biological process thanks to the unmatched sensitivity of our unique single photon detection technology - 37 photons/s/cm²/sr.
- Quantify with the greatest accuracy by monitoring the signal kinetics enabled by our real - time acquisition.
- Discover unique insights by visualizing moving animals.
- Build your own customized system for your experimentation needs.
- Improve animal welfare by applying non-invasive procedures.
NIR II / SWIR Imaging
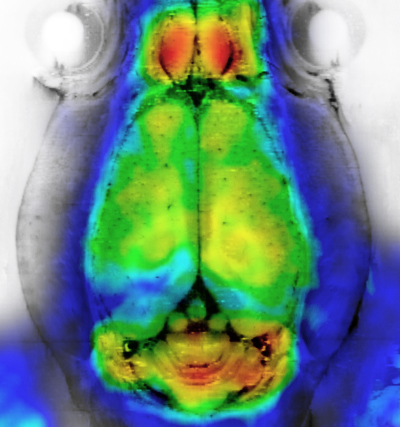
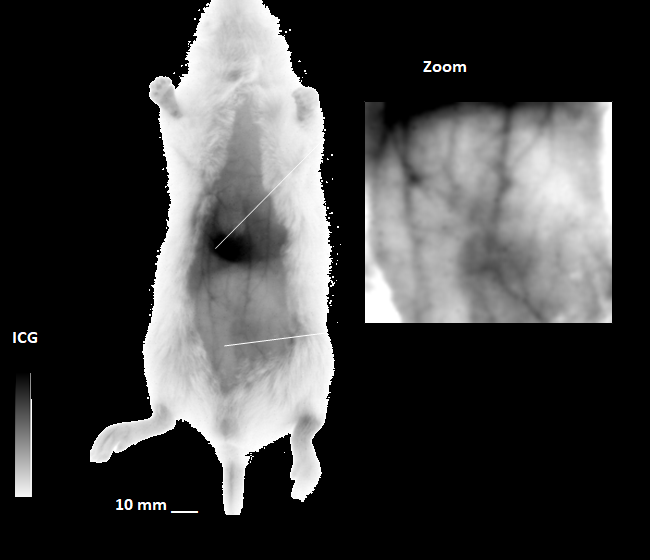
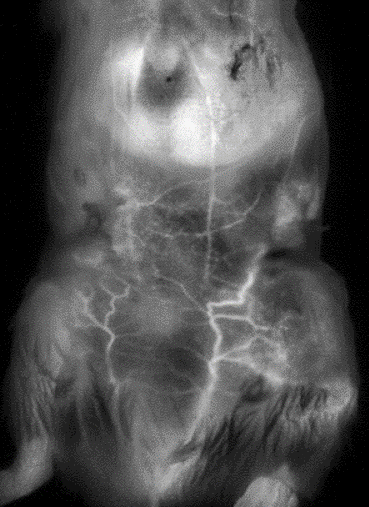
Our unique 2D / 3D short-wavelength region (SWIR) imaging system dramatically decreases blood, tissue and organ autofluorescence, resulting in lower light absorption and / or scattering. As a result, you can now acquire images with deeper penetration, higher sensitivity, and higher spatial resolution.
With our SWIR system, you will be able to:
- Achieve full tissue transparency and obtain deep tissue information
- Minimize the number of invasive experiments for the observation of organs (including the brain) and vasculature.
- Study rapid biological processes in greater detail compared to classic imaging modalities.
- Customize your own system to respond to your specific application needs, with our latest nanothermometry option for instance.
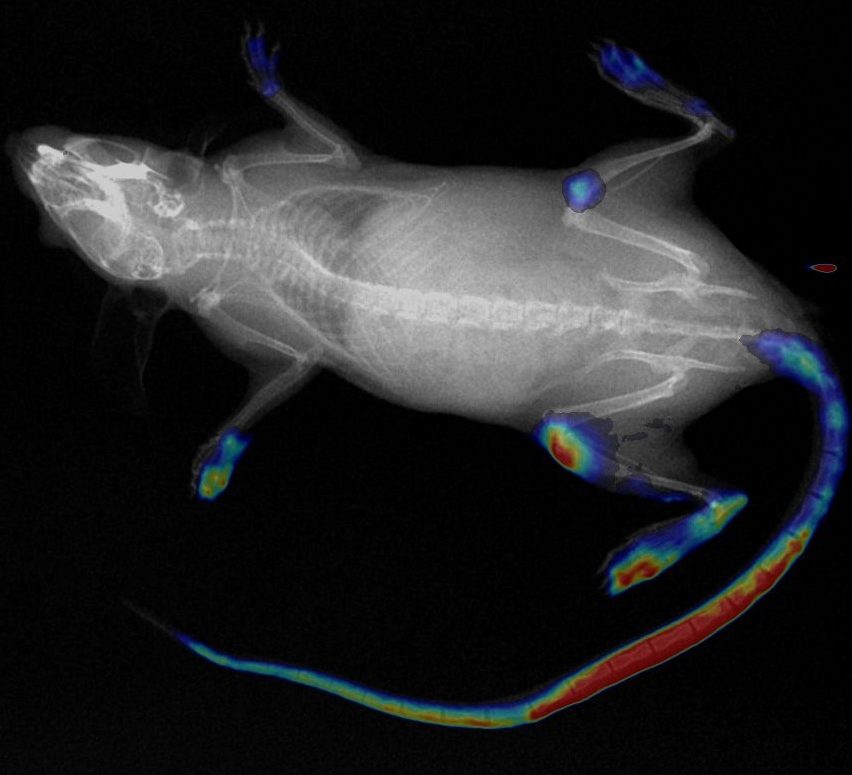
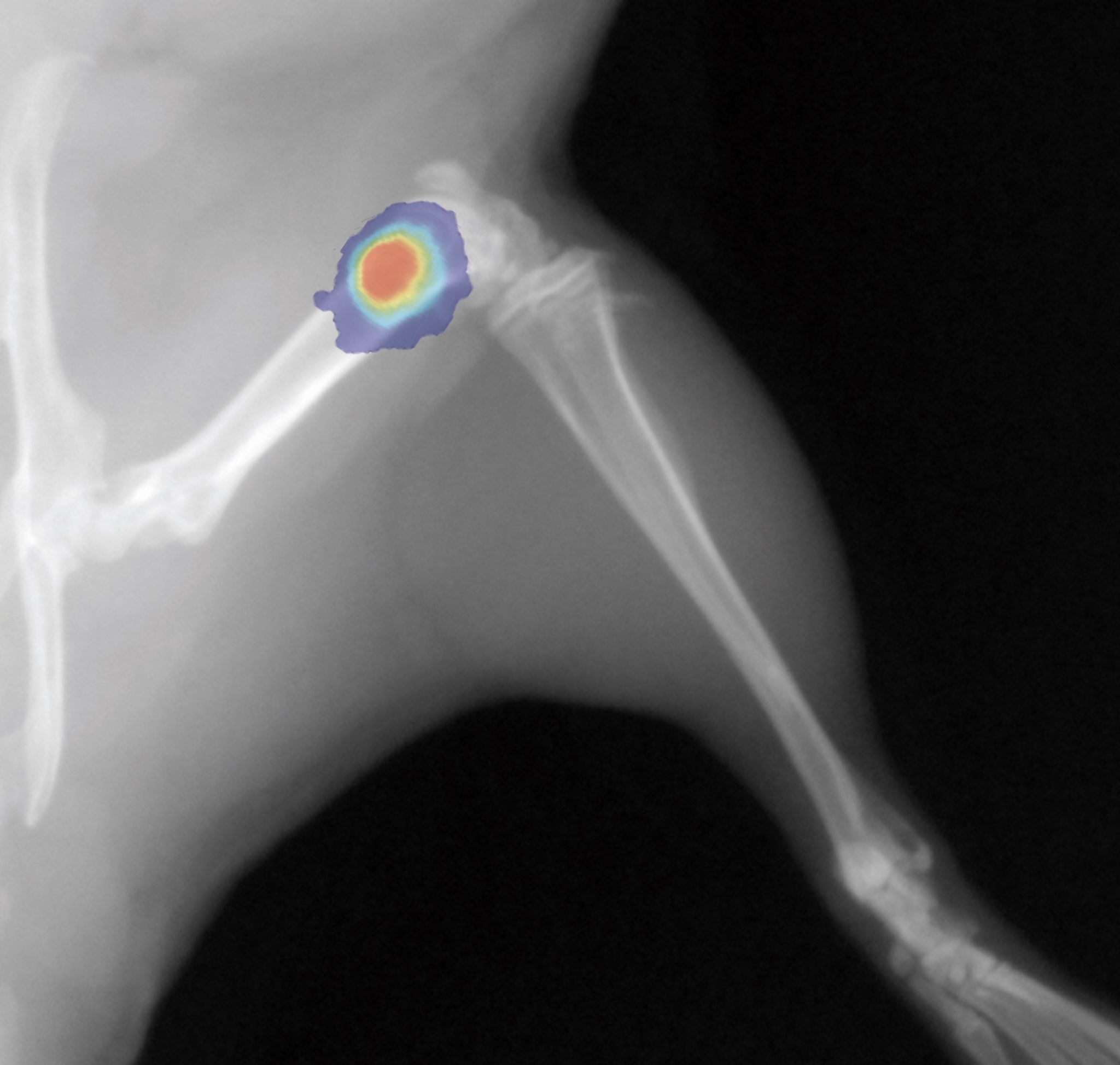
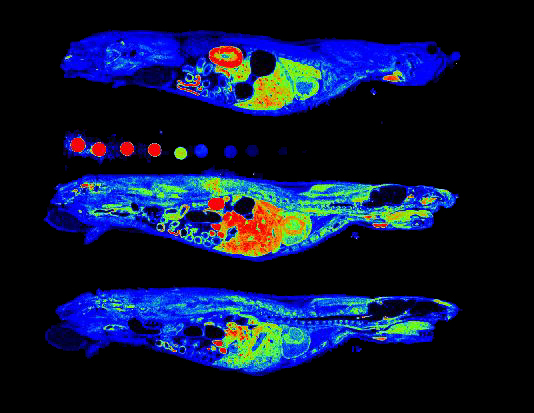
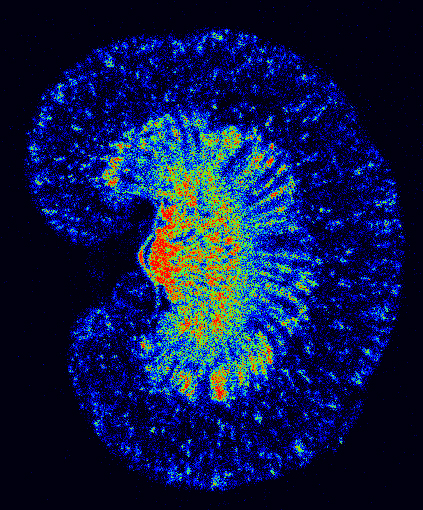
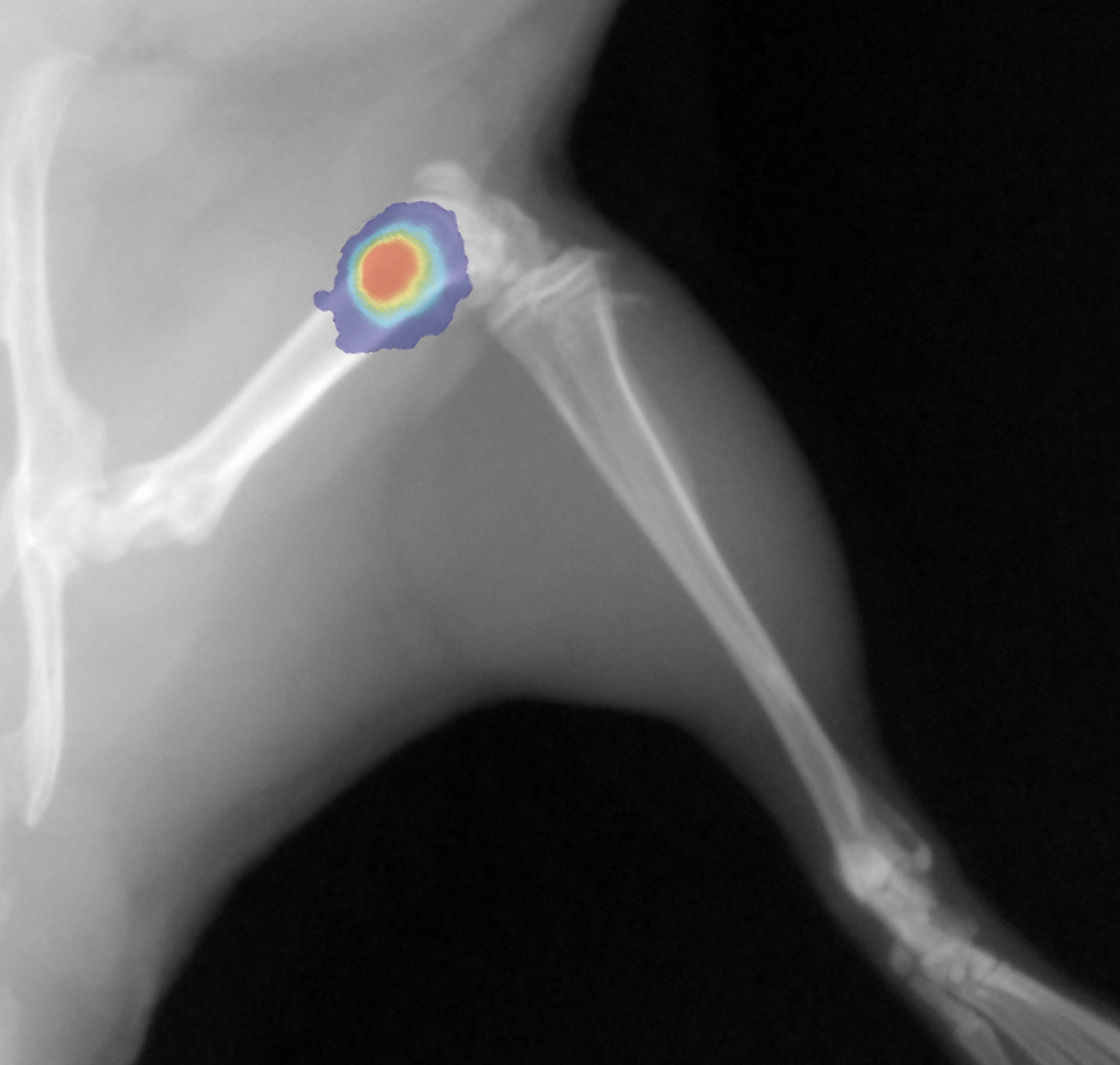
Autoradiography Imaging
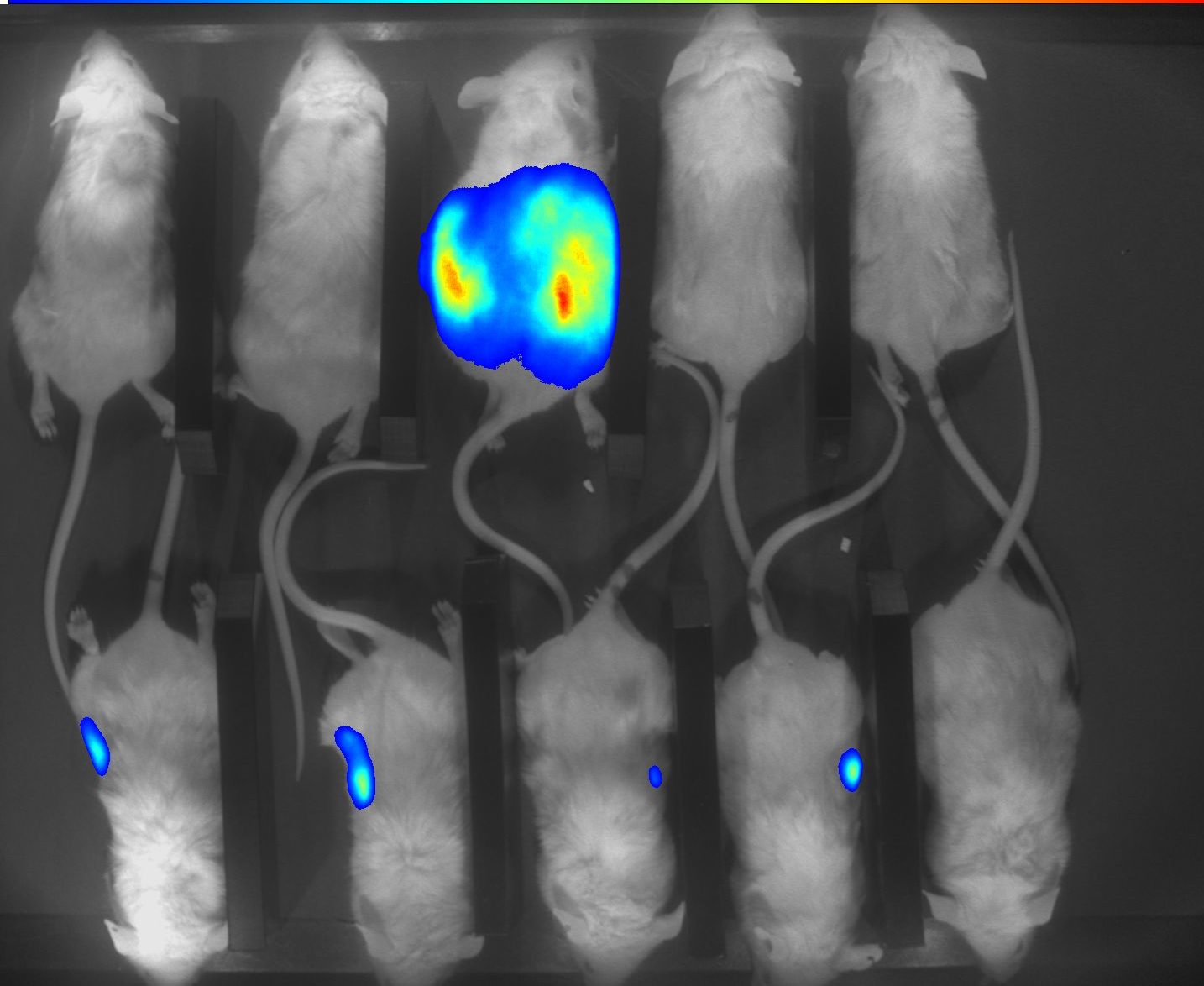
Our autoradiography systems enable you to verify and quantify the biodistribution of your injected radioisotopes with micrometric precision and high sensitivity.
Our digital autoradiography system leverages the gas detection technology invented by our founder Georges Charpak. Nobel Prize in Physics in 1992.
Depending on your research needs, we offer systems optimized for high throughput and high resolution.
With our autoradiography systems, you will be able to:
- Detect and quantify both high and ultra-low radioisotopes signals in a single data acquisition.
- Save hours or days by acquiring images in real-time.
- Avoid failed experiments caused by saturation.
- Use minimal amounts of radiolabeled compounds.
- Reduce radioactive waste and minimize experimental costs .

bioluminensce & fluorescence
Our real-time optical imaging systems enable you to see different types of complex biological processes by combining bioluminescence, fluorescence, X-ray imaging technologies in a single instrument.
Thanks to our modular approach, your specific requirements for optimized localization, resolution and visualization can be easily accommodated by adding 3D, macrolens, in action, X-ray options.
As a pioneer in imaging of non-anesthetized animals, Biospace Lab provides a unique solution to perform your experimentations on free-moving animals in physiological conditions.
With our systems, you will be able to:
- Detect the tiniest signals from a biological process thanks to the unmatched sensitivity of our unique single photon detection technology.- 37 photons/s/cm²/sr
- Quantify with the greatest accuracy by monitoring the signal kinetics enabled by our real - time acquisition.
- Discover unique insights by visualizing moving animals.
- Build your own customized system for your experimentation needs.
- Improve animal welfare by applying non-invasive procedures.




NIR II / SWIR IMAGING (new)
Our unique 2D / 3D short-wavelength region (SWIR) imaging system dramatically decreases blood, tissue and organ autofluorescence, resulting in lower light absorption and / or scattering. As a result, you can now acquire images with deeper penetration, higher sensitivity, and higher spatial resolution.
With our SWIR system, you will be able to:
- Achieve full tissue transparency and obtain deep tissue information
- Minimize the number of invasive experiments for the observation of organs (including the brain) and vasculature.
- Study rapid biological processes in greater detail compared to classic imaging modalities.
- Customize your own system to respond to your specific application needs, with our latest nanothermometry option for instance.


Autoradiography imaging
Our autoradiography systems enable you to verify and quantify the biodistribution of your injected radioisotopes with micrometric precision and high sensitivity.
Our digital autoradiography system leverages the gas detection technology invented by our founder Georges Charpak. Nobel Prize in Physics in 1992.
Depending on your research needs, we offer systems optimized for high throughput and high resolution.
With our autoradiography systems, you will be able to:
- Detect and quantify both high and ultra-low radioisotopes signals in a single data acquisition.
- Save hours or days by acquiring images in real-time.
- Avoid failed experiments caused by saturation.
- Use minimal amounts of radiolabeled compounds.
- Reduce radioactive waste and minimize experimental costs



Since 1989, Biospace Lab has been working in collaboration with prestigious academic centers and pharmaceutical laboratories to develop a unique selection of innovative preclinical imaging systems.

NANOTBTECH, H2020 program founded by European council
Novel and noninvasive way of measuring tissue temperature for oncology applications (micro-metastases) thanks to our unique SWIR imaging solution.

IN ACTIO, Project founded by Paris Region
Detect and quantify multiple bioluminescence signals on freely moving mice simultaneously. Follow neuronal biosensor and understand the social networking impact on Alzheimer pathology.

“The real-time acquisition of dynamic fluorescent signal curves from Biospace invivo imaging system, contribute to reconstruction visualized 3D tumor model, and provide intuitive and quantitative research methods, very helpful for research daily work”. The Institute of Automation, Chinese Academy of Sciences (CASIA), is one of the earliest national automation institutes in China, which was established in 1956.
Dr. Kun Wang, Associate Professor, CAS Key Laboratory of Molecular Imaging Institution of Automation Chinese Academy of Sciences, China
“Biospace Optima performs well in animal imaging, especially the high sensitivity and good signals”. The purpose of our institute is to train professional teachers and advanced researchers on Medical Physics, Biomedical Imaging, Radiobiology, Nuclear Medicine and Radiological Medical fields in clinical. Our department in addition to training professional personal is also specialized in research in the field of biomedical imaging and radiation sciences.
Dr Yi-Jang Lee Professor, Department of Biomedical Imaging and Radiological Sciences National Yang Ming University, Taiwan
“The PhotonIMAGER Optima has been a real asset in our experimental workflow by giving us the tools to detect and quantify both bioluminescence and fluorescence in our cancer mouse models. The PhotonIMAGER Optima has aided us in our research to monitor tumours in real-time over prolonged periods with minimal impact on the well-being of the mice. This device allows us to image up to six mice simultaneously, providing high resolution images along with an intuitive and powerful software package for downstream data analysis. We also recently purchased the ‘in actio’ module to increase our capabilities and provide imaging on non-anesthetised animals. This has been instrumental in maximising the outputs from our experiments and in aiding us to meet the principles of the 3Rs in animal research (Replacement, Reduction and Refinement). The customer service and training offered by Biospace Lab has been exemplary and their technical services are integral to the pursuit of our research goals”
Dr Patrick Hardinge, Dr Lee Parry, Dr Katie Stott and Dr Adam Higgins, European Cancer Stem Cell Research Institute, School of Biosciences, Cardiff University, UK

« CMMI, Center for Microscopy and Molecular Imaging, is a preclinical imaging platform equipped with many advanced equipments, such as PET, SPECT, CT, MRI, MSOT scanners for in vivo imaging. For optical imaging, the CMMI chose Biospace Lab at the end of the 2000s with the acquisition of the PhotonIMAGER RT. Biospace Lab has met our expectations by providing a system capable of performing BLI in real time, with very high sensitivity. Biospace Lab has also always provided quality technical and scientific support. Therefore, the CMMI has chosen to renew its confidence in Biospace Lab in 2018 with the acquisition of a new PhotonIMAGER Optima, which is even more efficient and modular. This device increased our imaging throughput by easily imaging 10 mice at a time, and provides high quality visible-NIR FLI. Adding new modules (MACROLENS, 4-views 3D and X-ray), enabled us to extend our imaging offer. In conclusion, Biospace Lab has been, and still is, an important partner in the development of our expertise in optical imaging. “
Dr. Lionel Larbanoix, laboratory manager of in vivo NiMI (Non-ionizing Molecular Imaging; laboratory head: Prof. Sophie Laurent), CMMI, Walloonia, Belgium.